Discovering How Nanoparticles Dissolve in Natural Water Environments
Published on by Water Network Research, Official research team of The Water Network in Academic
CEE researchers make breakthrough discovery that gold nanoparticles can dissolve when they interact with freshwater aquatic plants—instead of remaining stable as commonly thought.
By understanding these nano-bio interactions, we can use them to our benefit in designing better materials.
Source: Carnegie Mellon University
When you purchase anything from makeup to paint to sunscreen, chances are it contains engineered nanoparticles. These nanoscale materials have properties that are revolutionizing products—from medicine to agriculture to electronics. But eventually, those nanoparticles will reach natural environments. To use them safely and to their fullest potential, we need to know how they behave in real environments—and if that behavior leads to any unintended consequences.
Greg Lowry, professor of civil and environmental engineering at Carnegie Mellon University, studies how nanoparticles behave in and impact the environment. One way researchers have studied nanoparticle fate is by tracking gold nanoparticles—because they are stable and easy to find, or so researchers thought.
Recently, Lowry and Post-doctoral Researcher Astrid Avellan have made a breakthrough discovery: gold nanoparticles actually dissolve in freshwater environments, when they come into contact with mircroorganisms found on aquatic plants. During the process, gold ions are released, which will behave differently from the nanoparticles and could be toxic to some microorganisms.
The study did not measure toxicity so this doesn’t mean gold nanoparticles are harmful—instead, by better understanding their behavior in biologically active environments, scientists can ultimately use this knowledge to design better nanomaterials. Their findings weree published in Nature Nanotechnology.
“This study has opened our eyes to the importance of plants and the plant microbiome in determining the fate of engineered nanomaterials in freshwater environments,” said Lowry. “These plants, and biofilms in general, are important sinks for nanomaterials and are a fascinating compartment to study.”
The team looked at exactly what causes this transformation and how quickly it occurs. They conducted their tests in what is called a mesocosm—a controlled natural freshwater environment. The mesocosm, housed at the Center for Environmental Implications of NanoTechnology at Duke University, contains soil, sediment, water, plants, insects, fish, and microorganisms that ordinarily live in these natural environments.
Avellan and the research team released gold nanoparticles into the mesocosm water in very low amounts every week to mimic long-term, low dose inputs expected from nanomaterial uses.
They wanted to see how the nanoparticles would behave in a complex, biologically active ecosystem. After six months they found that 70% of the gold was accumulating with the aquatic plants, and that all of the gold nanoparticles had dissolved and changed to other forms of gold.
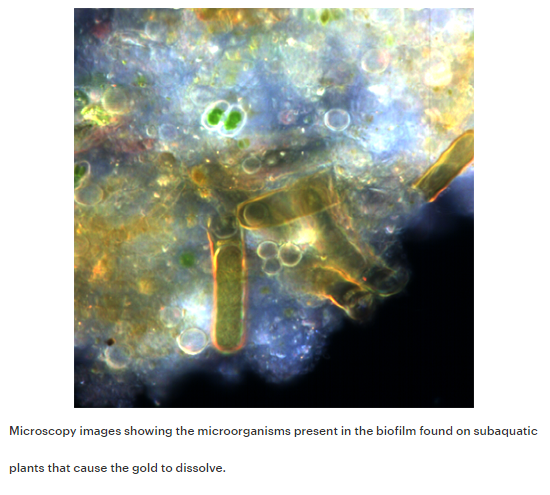
When they took a closer look at the biofilm, or a sticky substance made up of bacteria and microorganisms found on plants, they found that the microorganisms released cyanide that was interacting with the gold nanoparticles. The gold nanoparticles dissolved (or ionized) and formed gold-cyanide along with other gold complexes that remained with the plants.
Read full article: Carnegie Mellon University
Attached link
http://www.youtube.com/embed/K7U6qYwdN6sMedia
Taxonomy
- Metals
- Decontamination
- Heavy Metal Removal
- Decontamination
- Nano Materials
- Contaminant Control
- Water filtration, purifiers, nano filtration