PFAS chemicals do not last foreverOnce dubbed "forever chemicals," per-and polyfluoroalkyl substances, or PFAS, might be in the market for a new...
Published on by Trudi Schifter, CEO and Founder AquaSPE
Once dubbed "forever chemicals," per-and polyfluoroalkyl substances, or PFAS, might be in the market for a new nickname.
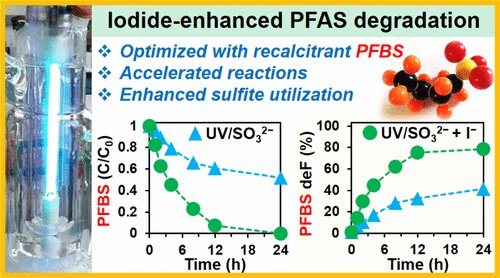
That's because adding iodide to a water treatment reactor that uses ultraviolet (UV) light and sulfite destroys up to 90% of carbon-fluorine atoms in PFAS forever chemicals in just a few hours, reports a new study led by environmental engineering researchers at UC Riverside. The addition of iodide accelerates the speed of the reaction up to four times, saving energy and chemicals.
"Iodide is really doing some substantial work," said corresponding author Jinyong Liu, an assistant professor of chemical and environmental engineering. "Not only does it speed up the reaction but it also allows the treatment of a ten times higher concentrations of PFAS, even some very recalcitrant structures."
Liu's lab has been working on ways to destroy PFAS through photochemical reactions since 2017. The new method has already attracted interest from industry and Liu's group is partnering with companies to conduct pilot tests.
Synthetic chemicals known as PFAS contain multiple very strong carbon-fluorine bonds. Widespread use of these nonbiodegradable compounds in countless products since the 1940s has contaminated water supplies across America, with various negative health effects on human and animals. Because the carbon-fluorine bond is very hard to break, PFAS pass through most water treatment systems unchanged.
Photochemical degradation by UV light and sulfite (SO32−) is, to date, one the most effective ways to break PFAS down. The original process used a lot of electricity because the chemical reactions occurred slowly. It also left multiple carbon-fluorine bonds remaining in the degradation products, with unknown health effects.
Last year, the researchers reported that oxidation treatments before and after the UV/sulfite treatment can achieve almost 100% destruction of carbon-fluorine bonds in various major PFAS pollutants.
Attached link
https://phys.org/news/2022-05-pfas-chemicals.htmlTaxonomy
- PFAS